AI has moved from a speculative topic in healthcare boardrooms to an operational infrastructure layer. Among the models competing for this space, Anthropic's Claude has emerged as a technically distinctive option — one whose architecture, safety guarantees, and rapidly expanding native integrations have made it a compelling choice for organizations operating under the regulatory and clinical pressures unique to healthcare. This guide synthesizes the state of Claude for Healthcare as of May 2026: what Anthropic has actually built, what it performs like in clinical benchmarks, who is deploying it and how, and what implementation teams need to understand before they start.

Claude for Healthcare at J.P. Morgan 2026
In January 2026, during the J.P. Morgan Healthcare Conference, Anthropic announced Claude for Healthcare — a dedicated set of tools and integrations designed to connect Claude's reasoning capabilities with the data infrastructure that US healthcare organizations actually run on. The announcement marked a significant strategic escalation: rather than offering a general-purpose model that healthcare organizations could adapt, Anthropic is now delivering purpose-built connectors, skills, and compliance structures designed specifically for clinical and administrative use cases.
The launch was not Anthropic's first move into the life sciences space. In October 2025, the company had released Claude for Life Sciences, which focused primarily on research and pharmaceutical applications. Claude for Healthcare extends that foundation into clinical operations, revenue cycle management, and patient-facing workflows — areas where the stakes, the regulatory complexity, and the volume of manual administrative work are highest.
Notably, the announcement came less than 24 hours after OpenAI unveiled its own healthcare-focused offering, signaling that the competition for AI infrastructure in healthcare has moved from concept to direct product rivalry.
What Claude for Healthcare Actually Includes?
Claude for Healthcare is best understood not as a single product but as a layered set of capabilities — native integrations to medical databases, a HIPAA-compliant infrastructure tier, and specialized skills for clinical and administrative workflows. Understanding each layer is essential for accurate scoping of what any implementation will and will not handle.
CMS Integration and Prior Authorization
One of the most operationally significant components of the Claude for Healthcare launch is a native connector to the Centers for Medicare & Medicaid Services (CMS) Coverage Database. This integration allows Claude to pull payer coverage requirements in real time, verify local coverage determinations, and check prior authorization criteria against patient clinical data — all within a HIPAA-eligible environment.
The practical application is substantial. Prior authorization represents one of the most labor-intensive and error-prone workflows in US healthcare. The American Medical Association's annual survey has consistently found that the process consumes more than fourteen physician hours per week in a typical practice. With the CMS connector, Claude can automatically surface coverage requirements for a given procedure or medication, check clinical criteria against patient records, and propose an authorization determination with supporting documentation — leaving the final decision to the reviewing clinician or administrative staff. This is a human-in-the-loop model: Claude augments the process, it does not replace the oversight.
ICD-10 and CPT Medical Coding Skill
A dedicated coding skill allows Claude to map physician notes and clinical documentation to ICD-10 diagnostic codes and CPT procedure codes automatically. Rather than requiring coders to manually extract billable diagnoses from often lengthy and unstructured clinical notes, Claude analyzes the documentation and proposes a coding assignment with reasoning — which a coder can then review, modify, and approve.
The revenue cycle implications are meaningful. Coding errors — whether undercoding that leaves revenue unrealized or upcoding that creates compliance exposure — are a persistent source of financial leakage in healthcare organizations. Automating the first-pass coding step with a model that can reason across complex clinical language reduces both error rates and the time coders spend on straightforward cases, freeing capacity for the complex encounters that genuinely require human expertise.
EHR Integration via HealthEx
Through a partnership with HealthEx, Claude now has access pathways to patient health records stored in Apple Health and Android Health Connect. This integration enables patients — with appropriate consent — to connect their personal health data and ask Claude natural-language questions about their own medical history, lab results, and care trajectories.
For health systems building patient engagement infrastructure, this represents a meaningful capability: AI-assisted navigation of personal health data that can reduce the burden on clinical staff for routine informational queries while improving patients' understanding of their own conditions. The HealthEx partnership routes this access through HIPAA-eligible infrastructure, maintaining the compliance posture required for clinical deployment.
PubMed and Scientific Literature Integration
For life sciences applications, Claude's native integrations extend to PubMed and 10x Genomics datasets. New skills allow Claude to search scientific literature, identify contradictions across published studies, and generate testable research hypotheses complete with full citations. This moves the model beyond summarization toward genuine scientific synthesis — a capability with obvious applications in pharmaceutical R&D, where the volume of published literature consistently outpaces any individual researcher's ability to stay current.
Microsoft Foundry Integration
For organizations running on Microsoft's cloud infrastructure, Claude is now available through Microsoft Foundry, providing a deployment path that fits within existing Azure-based healthcare architectures. This integration means healthcare organizations do not need to build new cloud infrastructure to access Claude — they can deploy through the Microsoft environment their compliance and security teams have already reviewed and approved.
Clinical Benchmarks: What the Research Shows About Claude For Helathcare?
Deploying AI in clinical environments demands a higher standard of evidence than most enterprise software decisions. The benchmark data available as of 2026, drawn from academic publications and Anthropic's own reported evaluations, provides a useful but incomplete picture of where Claude performs well and where caution remains warranted.
MMLU Medical Performance
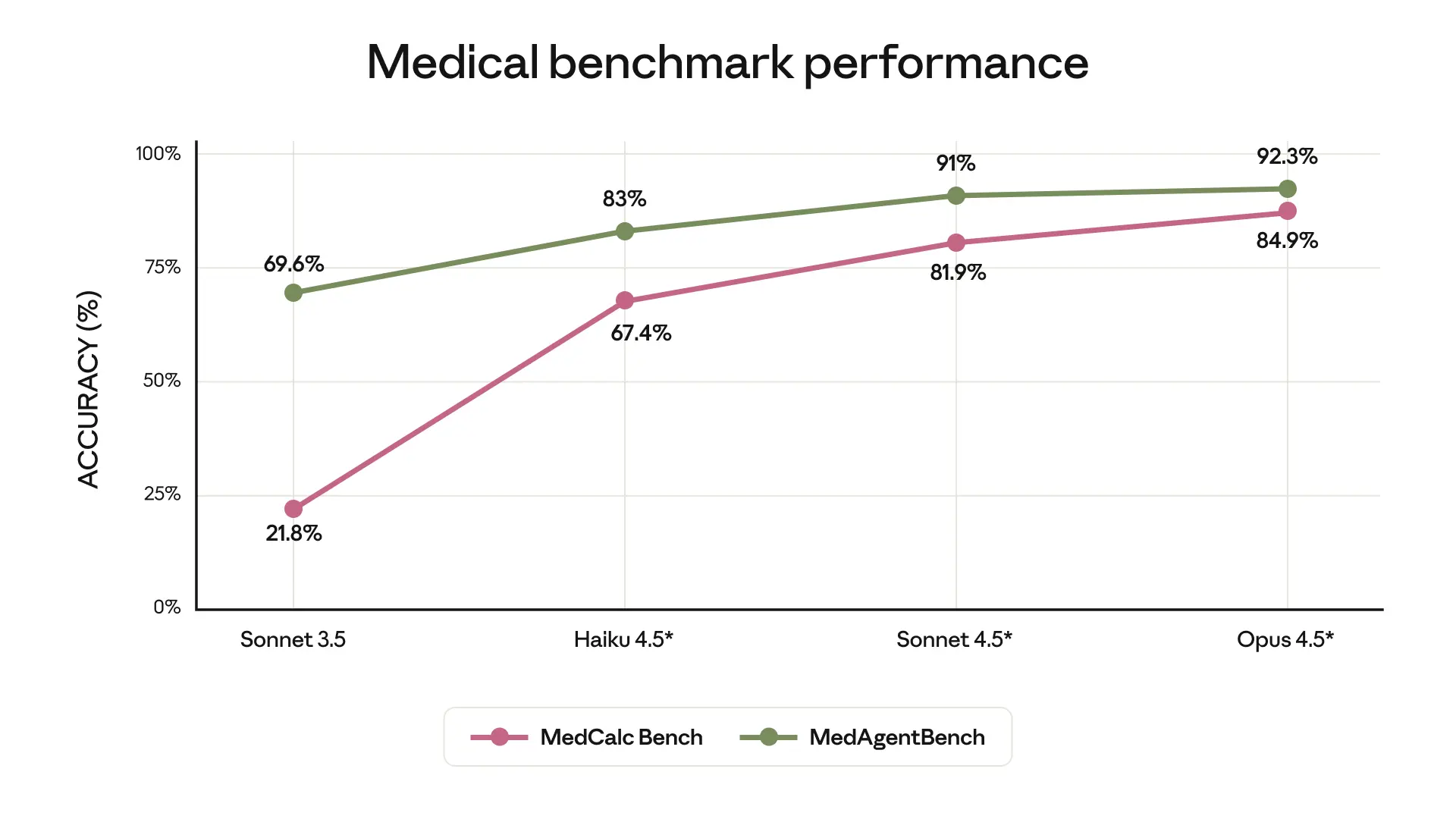
Research published in JAMA Network Open (April 2026) evaluated Claude 3.5 Sonnet and Claude 4.5 Opus on the MMLU medical and biology subsets. Both models achieved scores above 90%, placing them in a performance tier that exceeds many practicing physicians on standardized knowledge assessments. These results are consistent with Anthropic's own reported benchmarks and with independent evaluations conducted by academic medical centers.
The researchers noted an important nuance, however: while Claude performed well at reaching a final diagnosis, it demonstrated a tendency toward conservatism in deciding which next diagnostic steps to recommend. In other words, the model is strong at diagnostic reasoning but more cautious than clinicians in recommending specific follow-up tests. Whether this conservatism is a feature or a limitation depends entirely on the clinical context. For a decision-support tool in a resource-constrained environment, clinical conservatism may be appropriate. For an application designed to accelerate diagnostic workup, it may require calibration.
Multi-Step Clinical Reasoning and Self-Correction
In internal evaluations, Anthropic has documented Claude's capacity for extended reasoning chains — the model can work through more than 100 reasoning steps when it detects an inconsistency between its initial diagnostic conclusion and the underlying laboratory data. This self-correction capability is particularly relevant for complex cases where the clinical picture evolves as new information becomes available. A model that can recognize and revise an earlier conclusion when confronted with contradictory evidence behaves more like a careful clinician than a pattern-matching classifier.
This extended reasoning also reduces the hallucination risk that has made clinical AI deployment contentious. Claude's Constitutional AI architecture is designed to make the model acknowledge uncertainty rather than generate confident-sounding responses when the underlying evidence is weak — a property that Banner Health's CTO, Michael Reagin, cited explicitly when explaining why the organization chose Claude for its enterprise deployment.
Real-World Deployments: Who Is Using Claude and How
Banner Health: BannerWise at Scale
The most extensively documented real-world Claude for Healthcare deployment is BannerWise, the AI assistant built by Banner Health — a 33-hospital system based in Arizona with $15.6 billion in annual revenue and operations across six states. Banner deployed BannerWise to more than 55,000 employees in late 2025, making it one of the largest enterprise-wide AI rollouts in US healthcare to date.
The initial clinical use case that drove adoption was oncology chart summarization. Manually summarizing an oncology patient record — which can run to hundreds of pages — required approximately eight hours of physician time per patient. With Claude handling the initial summarization, that process is automated, with the physician reviewing and validating the AI-generated summary. By the end of 2025, BannerWise had processed more than 1,400 clinical notes, with 80 to 85 percent of users reporting time savings and improved documentation accuracy.
Banner's CTO, Michael Reagin, has been direct about what drove the selection: the model's documented tendency to acknowledge what it does not know, rather than generating plausible-sounding but incorrect answers. In a clinical environment where a confident hallucination could influence a care decision, this property is not a technical nicety — it is a safety requirement.
Novo Nordisk and Genmab: Life Sciences R&D
Novo Nordisk and Genmab have been identified as early adopters of Claude's life sciences capabilities, using the model to support pharmaceutical research workflows. These implementations focus on literature synthesis, hypothesis generation, and the analysis of genomic and chemical datasets at a scale that would be impractical through manual research processes.
Novartis: Preclinical Toxicity Prediction
Novartis, working through the Axiom platform, is applying Claude to predict drug toxicity at the preclinical stage. The model analyzes billions of tokens of genomic and chemical data to identify toxicity signals before compounds enter expensive clinical testing phases. Accelerating preclinical screening has long been one of the clearest ROI opportunities in pharmaceutical R&D — the costs of identifying a toxic compound after Phase I trials are orders of magnitude higher than catching it in preclinical analysis.
Clinical Trial Protocol Design
In clinical trial design, Claude is being applied to help optimize study protocols — particularly endpoint selection for complex conditions like Parkinson's disease. The goal is to select trial endpoints that are both clinically meaningful and aligned with regulatory expectations, improving the probability of approval without compromising scientific validity. This is a domain where the combination of deep literature synthesis, regulatory knowledge, and structured reasoning across complex data is genuinely differentiated from traditional decision-support tools.
HIPAA Compliance: The Details That Matter
For any healthcare organization evaluating Claude, HIPAA compliance is not a checkbox item — it is the prerequisite that determines whether a deployment is legally permissible. Anthropic updated its Trust Center in conjunction with the Claude for Healthcare launch, and the details are specific enough to warrant careful reading.
Which Products Are HIPAA-Eligible
Only two Anthropic products are covered by a Business Associate Agreement (BAA): the Enterprise plan of Claude.ai and the native (1P) Anthropic API. Consumer products — Claude Pro, Claude Team — are explicitly excluded. Organizations processing Protected Health Information (PHI) through consumer-tier products are not HIPAA-compliant, regardless of any other security measures they have in place. This is not a gray area: it is a binary distinction with direct legal consequences.
Third-party platforms that offer Claude through their own infrastructure (AWS Bedrock, Google Cloud Vertex AI, Microsoft Foundry) operate under separate BAAs with Anthropic. Organizations using Claude through these platforms need to verify the compliance posture of the platform, not Anthropic's, as the relevant regulatory coverage.
Zero Data Retention (ZDR)
As of April 2026, Anthropic's native API is classified as an Eligible Service under its BAA without requiring a Zero Data Retention configuration. This change — from the previous requirement that ZDR be explicitly enabled for BAA coverage — simplifies the compliance posture for organizations that need to maintain logs and audit trails of AI interactions within their systems. Healthcare organizations can now implement standard logging and auditing practices alongside Claude deployments without compromising their BAA coverage.
Claude Code and Hospital IT Development
Anthropic has also clarified the compliance status of Claude Code, its programming assistant tool. Certain Claude Code functions are now covered under the BAA, which means hospital IT teams can use Claude Code to build internal healthcare applications within a compliant framework. This opens a significant path for health systems that want to develop custom integrations — EHR connectors, workflow automation tools, internal analytics applications — using AI-assisted development without stepping outside their compliance boundary.
Same Fight. Different Weapons: The Big Four Battle for US Healthcare
Q1 2026 produced something unprecedented in enterprise software: four of the world's most powerful technology companies — Amazon, Anthropic, Microsoft, and OpenAI — each launched a dedicated healthcare AI platform within a span of less than three months. The competitive window was January 7 through March 5, 2026. The target is the $4.5 trillion US healthcare market, where administrative costs alone exceed $360 billion annually and where AI-driven automation is now the primary lever available to health systems fighting shrinking reimbursement margins.
These four players are not entering from the same position, and they are not targeting the same workflows. Amazon arrives with EHR-native infrastructure and deep penetration into medical coding. Microsoft enters already embedded inside more than 50,000 hospitals through its M365 footprint and the Epic relationship. OpenAI is moving to own the enterprise policy layer — the discharge templates, prior authorization workflows, and documentation standards that define how clinical decisions get translated into billable events. And Anthropic is competing on the strength of its reasoning architecture and its claims and appeals knowledge base, the most legally complex and financially consequential corner of revenue cycle management.
For healthcare organizations, the practical question is not which platform is best in the abstract. It is which platform addresses the specific workflow gaps costing the organization the most — and which deployment model fits within existing infrastructure agreements, compliance postures, and IT capacity. The comparison below maps each player to the workflows they hit hardest, how they are priced, and what their entry into the market actually means for RCM operations.
Reading this map from a Flobotics perspective, the most consequential observation is not which platform is technically superior — it is how different the revenue implications are depending on where in the RCM cycle an organization is losing money. If front-end eligibility and intake coding is the primary source of denials, Amazon's EHR-native approach and Medical Coding SDK represent the most direct path to improvement. If the problem is back-end: denials working, appeals management, and policy determination — the workflows where clinical and legal reasoning intersect — Claude's architecture and its native CMS connector make it the strongest fit. Microsoft's Dragon Copilot is the right choice for organizations whose primary gap is ambient documentation and order entry efficiency, particularly those already standardized on Epic within an M365 environment. And OpenAI's ChatGPT for Healthcare is best evaluated as an enterprise policy layer tool — strongest where the goal is standardizing the templates and workflows that translate clinical documentation into billing-ready output at scale.
Implementation Priorities: Where the ROI Is
For organizations planning a Claude for Healthcare deployment, the current evidence base points clearly to two areas where the return on investment is most demonstrated and most near-term.
Revenue Cycle Management — specifically prior authorization automation and medical coding — offers the clearest financial case. The labor costs associated with prior auth processing, the denial rates driven by coding errors, and the administrative burden on clinical staff are all well-documented, well-measured, and directly addressable through Claude's native integrations. Organizations that have implemented comparable automation in these workflows consistently report denial rate reductions of 15 to 30 percent within 12 to 18 months.
Life Sciences R&D — particularly genomic data synthesis, literature review, and preclinical analysis — offers the largest magnitude of potential impact, though the implementation complexity and timeline are correspondingly greater. For pharmaceutical organizations, the compounding effect of accelerating any stage of the drug development pipeline is substantial.
The critical first step for any organization, regardless of use case, is the same: move to Enterprise API, execute the BAA, and establish the compliance infrastructure before any PHI touches the system. Everything else — the integrations, the workflow design, the change management — is built on that foundation.
This article draws on Anthropic's official announcements, published reporting from Fierce Healthcare, Becker's Hospital Review, TechCrunch, Fortune, and the Microsoft Cloud Blog, as well as research published in JAMA Network Open. Information reflects the state of Claude for Healthcare as of May 2026.









.png)


.png)

